This Application is currently in Beta Testing.
See How It Works
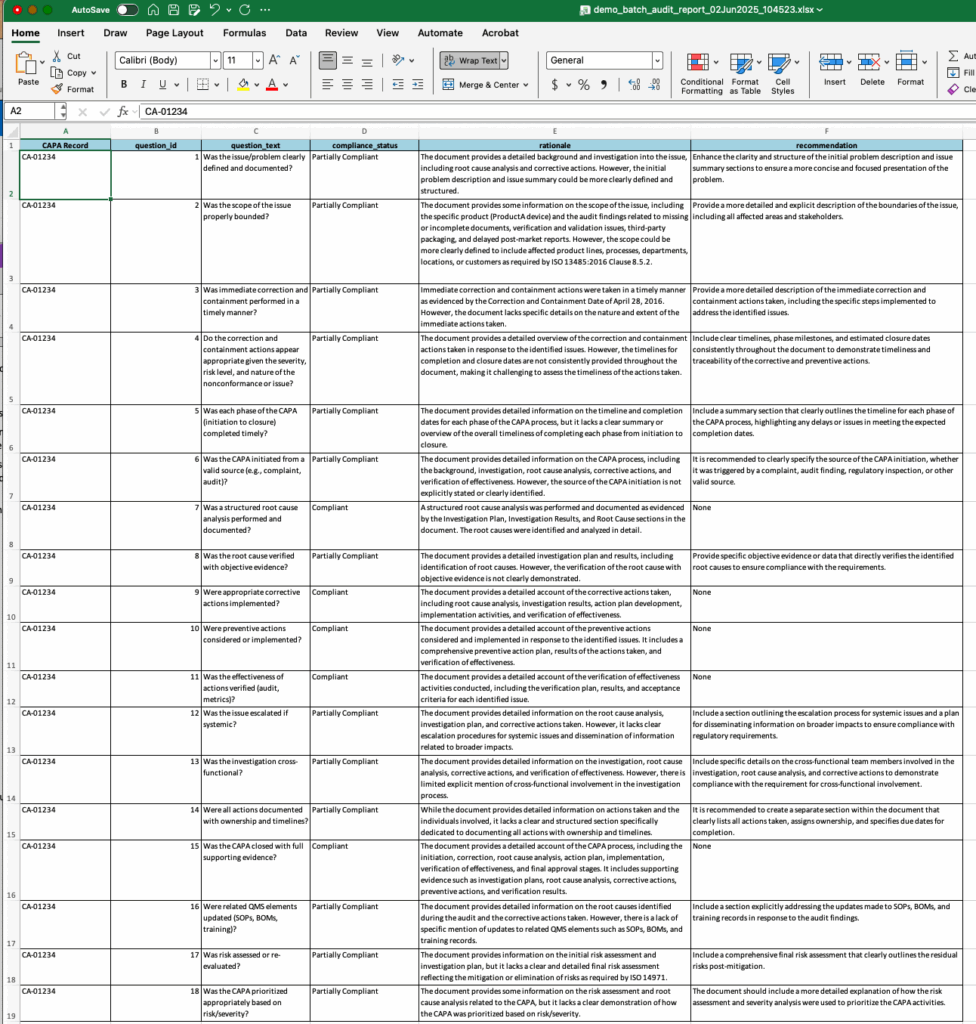
The CAPA Auditor applies FDA and ISO standards directly to your corrective and preventive action (CAPA) process, helping quality teams assess compliance, effectiveness, and documentation quality in minutes. Built with embedded logic from FDA 21 CFR Part 820 and ISO 13485, the tool uses 18 structured questions to evaluate each CAPA’s feasibility, completeness, and regulatory alignment. It delivers a detailed compliance scorecard with recommendations for remediation or process improvement.
ROI /Value
Manual CAPA reviews often take hours and vary in consistency across reviewers and sites. The CAPA Auditor standardizes this process, reducing review time by up to 80% while ensuring full traceability to regulatory expectations. It helps organizations close CAPAs faster, prepare for audits confidently, and strengthen long-term quality system performance.
Features
- Evaluates CAPAs using FDA and ISO 13485 criteria.
- 18-question framework assessing structure, root cause, action, and verification steps.
- Classifies responses as Compliant, Partially Compliant, or Not Compliant.
- Provides written rationale for each compliance rating.
- Generates actionable recommendations for improvement and closure readiness.
- Summarizes results in a visual compliance dashboard for quick review.
CAPA Auditor (BETA)